Welcome to the ATBC Study Web Site
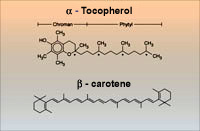
The Alpha-Tocopherol, Beta-Carotene Cancer Prevention (ATBC) Study was a cancer prevention trial conducted by the U.S. National Cancer Institute (NCI) and National Public Health Institute of Finland (which became the National Institute for Health and Welfare) from 1985 to 1993. The purpose of the study was to determine whether specific vitamin supplements prevented lung cancer and other cancers in a cohort of 29,133 male smokers in southwestern Finland. The 50-69 year old participants were randomized to receive one capsule daily (for five to eight years) containing one of the following: 50 milligrams vitamin E (as dlalpha-tocopheryl acetate), 20 milligrams of beta-carotene (a precursor of vitamin A), both vitamin E and beta-carotene, or a placebo (i.e., an inactive capsule that appeared identical to the vitamin capsules).
This web site is dedicated to the 29,133 participants in the ATBC Study who have contributed to our understanding of cancer etiology and prevention.
The participants stopped taking the vitamin supplements in April 1993. However, in order to evaluate the long-term effects of the vitamins on cancer incidence, and overall and cause-specific mortality, they were followed after the trial ended using data from the national registries in Finland. The researchers acquired additional data for cancer incidence and mortality related to specific causes through December 2012 and for total mortality through December 2013 (twenty years beyond the end of the trial).
For more information regarding study design and initial findings, download The alpha-tocopherol, beta-carotene lung cancer prevention study: design, methods, participant characteristics, and compliance and the The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers.
